Do Non FDA Approved Treatments Mean They Are Not Good?
Many patients feel worried when they hear that a treatment is not FDA approved. There is a common belief that if something is not FDA approved, it must be unsafe or ineffective. In aesthetic and regenerative dermatology, this is not always true.
Understanding this difference is important, especially with newer treatments like salmon DNA, exosomes, stem cells, and biologic injectables.
What FDA Approval Really Means
FDA approval is a regulatory process mainly designed for drugs and medical devices in the United States.
It means
The product has gone through large, expensive clinical trials
The indication is clearly defined
The manufacturer has applied for approval for that specific use
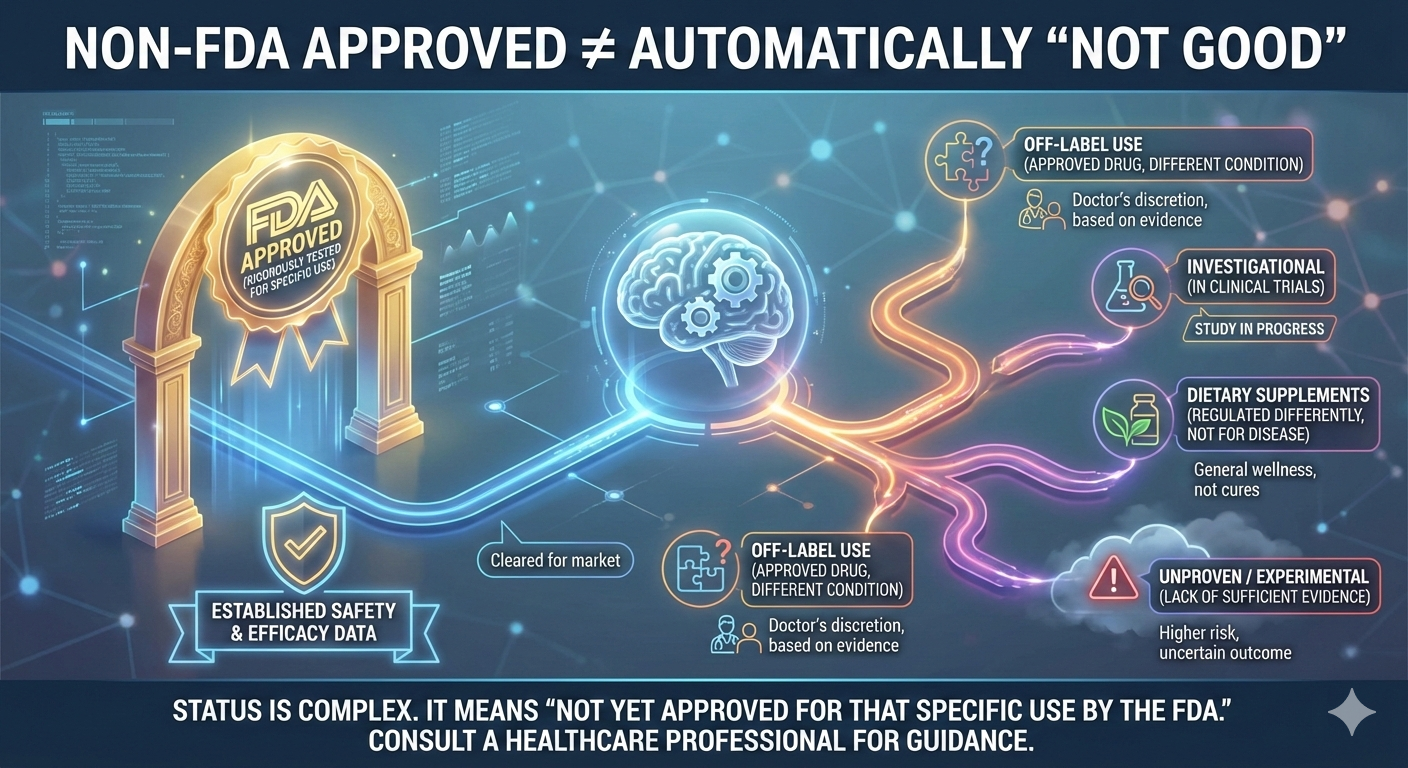
FDA approval does not automatically mean a treatment is superior.
Lack of FDA approval does not automatically mean a treatment is unsafe or useless.
Many effective treatments are widely used for years before or without FDA approval for aesthetic indications.
Why Many Aesthetic Treatments Are Not FDA Approved
Aesthetic medicine evolves faster than regulatory systems.
Common reasons include
The treatment is used off label
The company has not applied for FDA approval due to cost
The product is approved in Europe or Asia but not yet in the US
The treatment is classified as a biologic or regenerative therapy, which follows different regulations
For example
PRP was used for years before clear regulatory frameworks
Many fillers are used off label for areas not mentioned in FDA labeling
Energy devices often evolve faster than their approvals
Salmon DNA Injections
Salmon DNA or polynucleotides are widely used in Europe and Asia.
What we know
They improve hydration, elasticity, and skin repair
They work by stimulating fibroblasts and tissue regeneration
They are not fillers and do not add volume
They are supported by
Laboratory studies
Clinical experience
Published international data
Lack of FDA approval does not mean they are ineffective. It mainly reflects regulatory pathways, not poor science.
Exosome Therapy
Exosomes are signaling particles released by cells.
They work by
Reducing inflammation
Stimulating repair pathways
Improving collagen and skin quality
Why FDA approval is complex
Exosomes are biologics
Their classification varies by source and processing
Regulatory standards are still evolving
In clinical practice, properly sourced and well processed exosomes show promising results in skin rejuvenation, hair loss, and post procedure recovery.
Stem Cell Based Treatments
Most aesthetic stem cell treatments do not involve injecting live stem cells.
They usually use
Stem cell conditioned media
Growth factors
Cell derived signaling molecules
These treatments aim to activate the skin’s own repair mechanisms rather than replacing cells.
Again, regulation is complex, but growing scientific evidence supports their regenerative role.
FDA Approved Does Not Mean Zero Risk
Even FDA approved treatments can have side effects if
Used incorrectly
Used in the wrong patient
Overused or combined poorly
Safety depends more on
Proper patient selection
Correct technique
Quality of the product
Experience of the doctor
What Matters More Than FDA Approval Alone
When evaluating any treatment, better questions to ask are
Is there scientific rationale behind it
Is there clinical data or real world evidence
Is the product well manufactured and traceable
Is it being used by trained medical professionals
Is it suitable for this specific patient
How Modern Dermatology Uses These Treatments
In modern dermatology, regenerative treatments are used to
Improve skin quality from within
Support healing after procedures
Address inflammation and aging at a cellular level
They are not magic solutions. They are tools used carefully, ethically, and selectively.
Final Take Home Message
Non FDA approved does not mean bad.
FDA approved does not mean perfect.
Good medicine is about evidence, experience, safety, and individualized care.
At Cutiscity, treatments are chosen based on science, patient benefit, and responsible practice, not just labels.